Chemistry of Brown Carbon
Organic aerosol particles are generally white in color, and therefore they scatter visible solar radiation back to space. This scattering prevents visible solar radiation from reaching the Earth’s surface thus cooling the climate. Light-absorbing “brown carbon” aerosols counteract this effect due to their ability to absorb solar radiation thus warming the climate. The term “brown carbon” refers to aerosol particles that contain organic compounds, which are capable of absorbing near UV and visible light [101]. Brown carbon can be classified into primary, which is emitted directly by various sources, such as combustion, and secondary, which is made of compounds produced directly in the atmosphere by multi-phase chemistry [101]. Both types of brown carbon play an important role in the atmospheric environment by decreasing atmospheric visibility, deteriorating air quality in urban areas, and affecting regional climate. The goal of our research is to understand the chemical composition and chemical aging of various types of brown carbon aerosols.
Primary Brown Carbon
 The largest source of brown carbon in the atmosphere is the burning of biomass, such as wood and grass. The smoke produced during inefficient combustion of biomass is highly complex and has brownish color (as shown in the photograph of particulate matter collected on a filter during a Black Spruce controlled burn). We have been investigating the chemical composition of brown carbon with help of a high performance liquid chromatography (HPLC) coupled to photo diode array (PDA) and high resolution mass spectrometry (HRMS) detectors in collaboration with Prof. Julia Laskin and Prof. Alexander Laskin from Purdue University. Examples of our recent publications on biomass burning smoke relying on the HPLC-PDA-HRMS include Refs. [114, 123, 127, 135, 138, 151]. These studies have provided a comprehensive classification of light-absorbing BBOA organics, such as lignin-derived products, distillation products, nitroaromatics, and polycyclic aromatic hydrocarbons, based on fuel and combustion conditions. The largest source of brown carbon in the atmosphere is the burning of biomass, such as wood and grass. The smoke produced during inefficient combustion of biomass is highly complex and has brownish color (as shown in the photograph of particulate matter collected on a filter during a Black Spruce controlled burn). We have been investigating the chemical composition of brown carbon with help of a high performance liquid chromatography (HPLC) coupled to photo diode array (PDA) and high resolution mass spectrometry (HRMS) detectors in collaboration with Prof. Julia Laskin and Prof. Alexander Laskin from Purdue University. Examples of our recent publications on biomass burning smoke relying on the HPLC-PDA-HRMS include Refs. [114, 123, 127, 135, 138, 151]. These studies have provided a comprehensive classification of light-absorbing BBOA organics, such as lignin-derived products, distillation products, nitroaromatics, and polycyclic aromatic hydrocarbons, based on fuel and combustion conditions.
Publication [151] was our first comprehensive study of how BBOA responds to aging UV irradiation. BBOA samples were irradiated directly on filters with near-UV radiation, followed by extraction and comprehensive chemical analysis. Lifetimes of individual chromophores depended on the fuel type and the corresponding combustion conditions. While individual chromophores disappeared on a timescale of several days, the overall light absorption by the sample persisted much longer, presumably because the condensed-phase photochemical processes converted one set of chromophores into another without complete photobleaching, or from undetected chromophores that photobleached more slowly. These results indicate that photobleaching of BBOA particles by condensed-phase photochemistry is relatively slow compared to aging by other oxidative mechanisms.
In addition to burning biomass, we have also been doing experiments with brown carbon produced by burning typical urban materials. We determined the extent to which particulate matter from the burning of different urban materials can absorb sunlight [190, 200]. We showed that light-absorbing properties of this particulate matter change upon exposure to solar UV radiation by (1) decomposing light-absorbing compounds (photobleaching) or (2) producing stronger light-absorbing compounds from photooxidation of smaller molecules (photoenhancement). On average, photoenhancement is a more important effect resulting in increasing light absorption by the particulate matter after it travels through the air. This means that smoke particles emitted during WUI fires may have a strong effect on visibility and climate.
Secondary Brown Carbon from Ammonia and Amines
A while back [62], we observed that SOA produced by oxidation of d-limonene changes from colorless to orange-brown when exposed to ammonia, a common atmospheric pollutant. This type of browning chemistry is not limited to SOA produced from d-limonene - we have been able to quantify the mass absorption coefficients of brown carbon material produced by this mechanism for a number of SOA types [77]. The following images show this transformation for an aerosol filter sample:
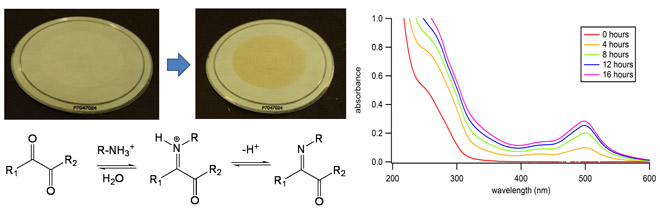
 The highly colored compounds produced by aging reactions are generated when ammonia reacts with carbonyl compounds present in the SOA. In collaboration with Prof. Julia Laskin and Prof. Alexander Laskin, we have been able to detect imine compounds that are likely responsible for the light absorption in aged limonene SOA using high resolution mass spectrometry [66, 99]. These compounds are similar to the products of Maillard reactions between sugars and amino acids that cause browning in food, for example, when toasting bread. Organic aerosols appear to get “toasted” on time scales of hours to days, which is a relevant atmospheric time scale. In collaboration with Purdue University, we investigated in detail the colored compounds in different secondary brown carbon species with HPLC-PDA-HRMS methods [109, 126]. We are also investigated the kinetics of reactions between selected dicarbonyls and nitrogen-containing compounds in order to better understand the reaction mechanism. Dicarbonyls that we have worked with include methylglyoxal [109, 126] and 4-oxopentanal [124]. We showed that brown carbon produced by reaction of methyl glyoxal and ammonium sulfate is highly photolabile and photobleaches on a time scale of minutes. The highly colored compounds produced by aging reactions are generated when ammonia reacts with carbonyl compounds present in the SOA. In collaboration with Prof. Julia Laskin and Prof. Alexander Laskin, we have been able to detect imine compounds that are likely responsible for the light absorption in aged limonene SOA using high resolution mass spectrometry [66, 99]. These compounds are similar to the products of Maillard reactions between sugars and amino acids that cause browning in food, for example, when toasting bread. Organic aerosols appear to get “toasted” on time scales of hours to days, which is a relevant atmospheric time scale. In collaboration with Purdue University, we investigated in detail the colored compounds in different secondary brown carbon species with HPLC-PDA-HRMS methods [109, 126]. We are also investigated the kinetics of reactions between selected dicarbonyls and nitrogen-containing compounds in order to better understand the reaction mechanism. Dicarbonyls that we have worked with include methylglyoxal [109, 126] and 4-oxopentanal [124]. We showed that brown carbon produced by reaction of methyl glyoxal and ammonium sulfate is highly photolabile and photobleaches on a time scale of minutes.
We found that browning chemistry is accelerated quite dramatically by evaporative cloud/fog processing of aerosols; the “brown carbon” compounds are produced much faster during evaporation of droplets containing dissolved SOA and ammonium sulfate [72, 124]. The fact that simple droplet evaporation can dramatically change the optical properties of the compounds dissolved in it has significant implications for direct forcing by aerosols.
We attempted to estimate the extent of conversion of ammonia into nitrogen-containing compounds using SOA chamber experiments [136]. These results were used to model the effect of this ammonia chemistry on particulate matter over the LA basin [128] and the entire US [129]. This project has culminated in publication [170], which reported results of difficult experiments to measure reactive uptake coefficient of ammonia (of the order of 10-5) and dimethylamine (from 10-5 to 10-4) on various types of SOA particles. Ammonia is more abundant in the atmosphere than amines, however, the larger observed reactive uptake coefficient suggests that amine uptake may also be a potential source of organic nitrogen in particulate matter. In Ref. [172], we used WRF-CMAQ to model reactive ammonia uptake by SOA in present time and for various future scenarios. The simulations including the NH3-SOA uptake under the warmer climate conditions of 2050 predicted a smaller impact of this chemistry on air quality compared to the current climate conditions.
Secondary Brown Carbon from VOC Photooxidation
Photooxidation of many VOCs, especially aromatic compounds, leads to light-absorbing SOA material. In one of the most cited papers from our group, we reported mass absorption coefficients for a number of types of SOA [107]. This was followed by more detailed studies of specific types of SOA to examine brown carbon produced by the photooxidation of toluene [174], naphthalene [93, 133, 187], and indole [119, 184]. For example, we measured the mass absorption coefficients and refractive indices for SOA prepared from indole under several different oxidation conditions [119, 184]. The most significant result was that the absorptive properties exhibited a strong dependence on relative humidity, highlighting the strong influence of water on the absorption coefficients of SOA from heterocyclic aromatic precursors. Indole SOA was strongly absorbing, which may contribute to degradation of visibility near its emission sources. We were able to unambiguously identify the main chromophore in the indole SOA using a combination of experiments and calculations [197]. In publication [187], we studied the effects of NH3, NOx, and relative humidity (RH) on the optical and molecular properties of naphthalene photooxidation SOA. Significantly, all of these parameters affected the characteristics of SOA, but in different ways. We found that NOx and RH control both molecular composition and optical properties, while NH3 has a stronger effect on composition and a minor effect on the absorption coefficient. We observed that certain combinations of parameters during SOA formation lead to effects on the composition and optical properties that cannot be anticipated from varying the two parameters separately.
Secondary Brown Carbon from Metal-Catalyzed Reactions
 In collaboration with Prof. Hind Al-Abadleh, we observed that reactions of dissolved Fe(III) from mineral dust particles catechol/guaiacol [105] and dicarboxylic acids [121] produce insoluble and highly light-absorbing polymeric particles. In publication [146], we showed that oxalate and sulfate, common ingredients of particulate matter, do not suppress the browning process. In publication [156] we demonstrated that the black organic matter coats the dust particles and affects the ability of these particles to nucleate ice. We also verified whether this chemistry takes place in more realistic mixtures of air pollutants. In a follow up work, we extended these measurements to various phenolic species [161] and to actual biomass burning smoke [182]. We observed the formation of highly-absorbing, insoluble particles when biomass burning organic aerosol was reacted with soluble Fe(III) under conditions characteristic of aerosol liquid water. Electron microscopy, done in collaboration with the group of Prof. Joe Patterson, revealed that the particles had a morphology reminiscent of soot particles and contained no detectable iron. These results suggest that light-absorbing aerosol particles can be produced from iron-catalyzed reactions in biomass burning plumes in the absence of photochemistry, with important implications for understanding the effects of biomass burning organic aerosol on climate. Publication [171] is a perspective article to encourage further research on this important topic. In collaboration with Prof. Hind Al-Abadleh, we observed that reactions of dissolved Fe(III) from mineral dust particles catechol/guaiacol [105] and dicarboxylic acids [121] produce insoluble and highly light-absorbing polymeric particles. In publication [146], we showed that oxalate and sulfate, common ingredients of particulate matter, do not suppress the browning process. In publication [156] we demonstrated that the black organic matter coats the dust particles and affects the ability of these particles to nucleate ice. We also verified whether this chemistry takes place in more realistic mixtures of air pollutants. In a follow up work, we extended these measurements to various phenolic species [161] and to actual biomass burning smoke [182]. We observed the formation of highly-absorbing, insoluble particles when biomass burning organic aerosol was reacted with soluble Fe(III) under conditions characteristic of aerosol liquid water. Electron microscopy, done in collaboration with the group of Prof. Joe Patterson, revealed that the particles had a morphology reminiscent of soot particles and contained no detectable iron. These results suggest that light-absorbing aerosol particles can be produced from iron-catalyzed reactions in biomass burning plumes in the absence of photochemistry, with important implications for understanding the effects of biomass burning organic aerosol on climate. Publication [171] is a perspective article to encourage further research on this important topic.
|


