Hygroscopicity on the Nanoscale: Water Uptake by Nanoparticles
Water-particle interactions play a very important role in the atmosphere. Every cloud and fog droplet has to grow on a pre-existing aerosol particle. The more hygroscopic are the particles the easier they take up water and grow into droplets.
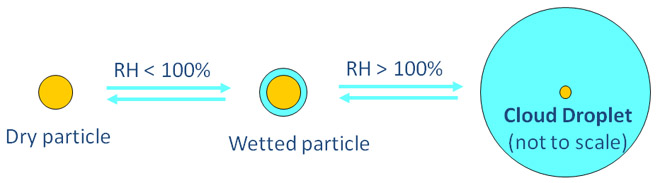
Nanoparticles with diameters less than 100 nm have unique hygroscopic properties because of their large surface-to-volume ratio. For example, the relative humidity at which NaCl undergoes wetted crystal-to-aqueous solution phase transition (called deliquescence) is about 75% for bulk NaCl crystals and for sufficiently large (> 100 nm) NaCl particles. However, the deliquescence relative humidity increases significantly for NaCl nanoparticles, primarily as a result of the Kelvin effect that makes water “uncomfortable” on highly curved surfaces.
 Our research focuses on the hygroscopic properties of multi-component ultrafine particles. Both the chemical content and particle diameter are critical factors that govern aerosol particle interaction with water; however, there is a lack of measurements of the hygroscopicity of multi-component nanoparticles. We combine experimental studies and modeling of hygroscopic properties of ultrafine aerosol composed of mixtures of soluble salts and organic surfactants. Hygroscopicity of such particles is of great fundamental interest; even a sub-monolayer of organic surfactants on the surface can significantly change the way nanoparticles interact with water. We perform measurements of water uptake by nanoparticles using a tandem differential mobility analyzer (a schematic diagram of our experimental setup is shown below). To aid in the interpretation of data, we do molecular dynamics and ab initio modeling of the interactions between the nanoparticles, surfactants and water. Our research focuses on the hygroscopic properties of multi-component ultrafine particles. Both the chemical content and particle diameter are critical factors that govern aerosol particle interaction with water; however, there is a lack of measurements of the hygroscopicity of multi-component nanoparticles. We combine experimental studies and modeling of hygroscopic properties of ultrafine aerosol composed of mixtures of soluble salts and organic surfactants. Hygroscopicity of such particles is of great fundamental interest; even a sub-monolayer of organic surfactants on the surface can significantly change the way nanoparticles interact with water. We perform measurements of water uptake by nanoparticles using a tandem differential mobility analyzer (a schematic diagram of our experimental setup is shown below). To aid in the interpretation of data, we do molecular dynamics and ab initio modeling of the interactions between the nanoparticles, surfactants and water.
Publication [59] was the first paper resulting from this project. It focused on the effects of the particle size and relative amount of surfactant AOT on the hygroscopic growth of mixed NaCl/AOT nanoparticles. For pure NaCl nanoparticles, the deliquescence relative humidity (DRH) increased as the particle size was decreased, in full agreement with previous measurements. Below the DRH the NaCl nanoparticles had an equivalent of 1-4 monolayers of water adsorbed on the surface. The addition of a sub-monolayer AOT coating reduced the DRH and suppressed the hygroscopic growth of the NaCl core. At AOT coverage levels exceeding one monolayer, a clear deliquescence transition was no longer discernible. A model assuming independent water uptake by AOT and NaCl failed to predict the observed growth factors of mixed AOT/NaCl nanoparticles reflecting a large contribution of the interfacial interactions between NaCl and AOT to the total free energy of the particles. There were clear indications that AOT/NaCl nanoparticles prepared by the electrospray aerosol source were enhanced in the relative mass fraction of AOT in comparison with the solution from which they were electrosprayed.
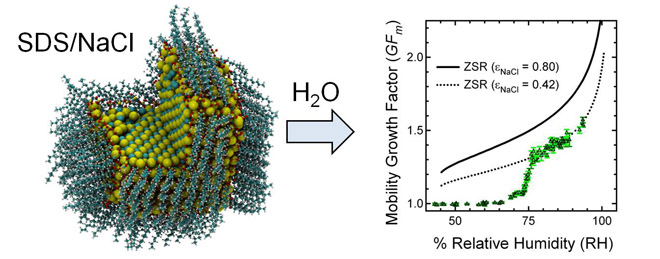
Our next publication [65] continued to investigate the properties of NaCl nanoparticles coated with surfactants. We used a combination of experimental and theoretical methodologies to explore water uptake on sodium chloride particles containing varying amounts of sodium dodecyl sulfate (SDS) to elucidate the interaction of water with well-defined, environmentally relevant surfaces. This study resulted in several key observations: 1) hygroscopic properties of nanoparticles are uniquely different from hygroscopic properties of the corresponding bulk mixtures with the same composition; 2) contrary to the intuitive expectations, the presence of an organic hydrophobic film makes the deliquescence occur at lower relative humidities (i.e., it helps adsorb water on the particle surface). These observations underscore the importance of interfacial properties in the phase transitions experienced by nanoparticles. Finally, we demonstrated [73] that nanoparticles produced by electrospray aerosol generators (EAGs) from ternary solutions are considerably enhanced in the more surface-active solutes. Specifically, surfactants tend to accumulate on surfaces of the particles during successive fission events in the electrospray source. This observation has practical ramifications not only for air-water interface science but also for aerosol nanotechnology and aerosol-assisted drug delivery, which rely on EAG as an instrument of choice for nanoparticle generation.
In collaboration with Dr. Theresa McIntire and with the group of Prof. Douglas Tobias, we explored the interactions of humid air with aerosols and thin films made of ionic liquids (ILs) [82,94]. Publication [82] was the first report of the interaction of water with aerosolized IL nanoparticles. The particles’ small size allowed the true IL-water vapor equilibrium to be achieved quickly. Growth curves for both ILs showed steady water uptake with increasing RH, without any evidence of phase transitions. Water vapor uptake by IL thin films was also examined using ATR-FTIR spectroscopy. Both experimental methods showed that [C2MIM][Cl] absorbs more water vapor than [C2MIM][BF4] over the entire RH range. Water molar fractions, calculated from growth curves, agreed well with those estimated from ATR-FTIR data. MD simulations, used to model IL-water interactions, revealed strong interactions between [Cl−] and water and considerably weaker interactions between [BF4−] and water. These experimental and theoretical data have provided a comprehensive picture of the behavior of absorbed water in ILs.
|


